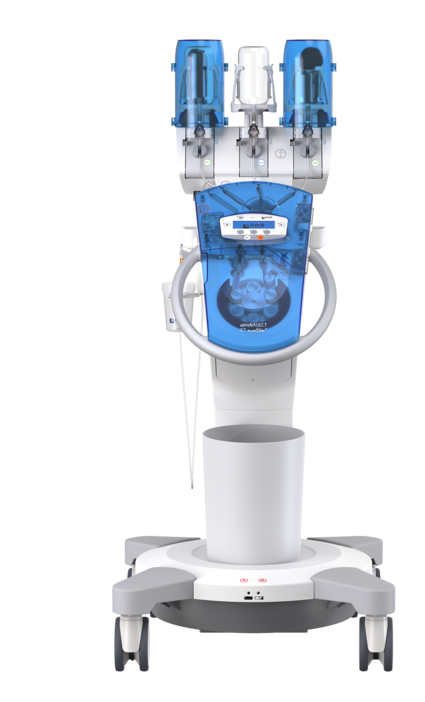
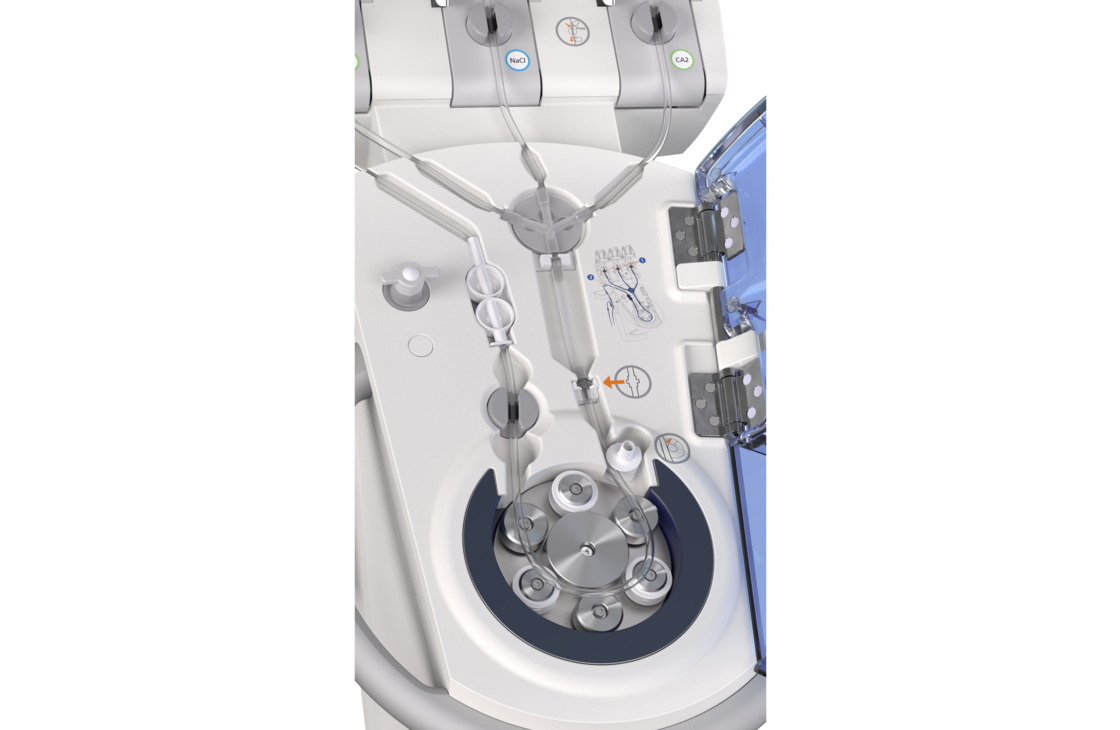
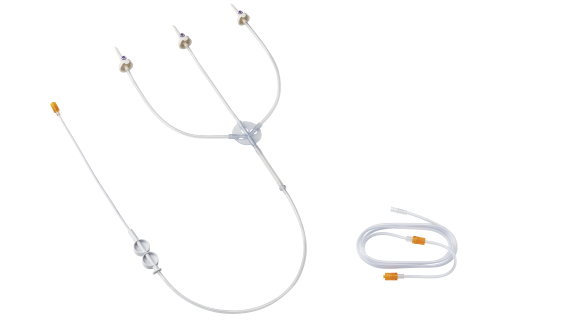
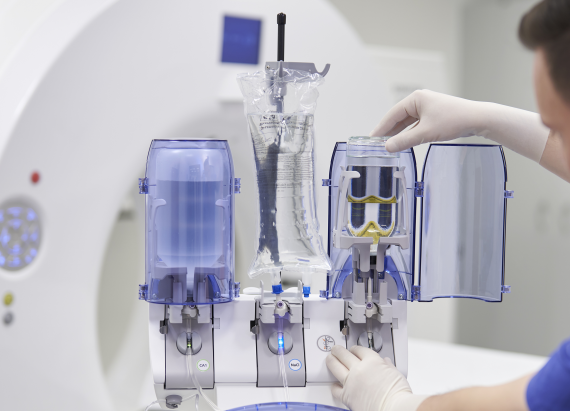
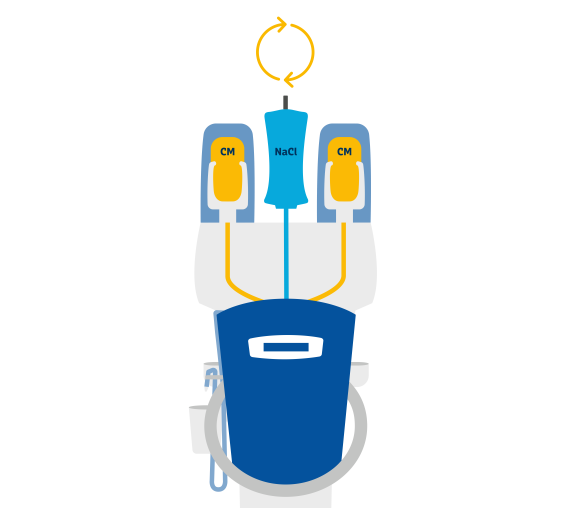
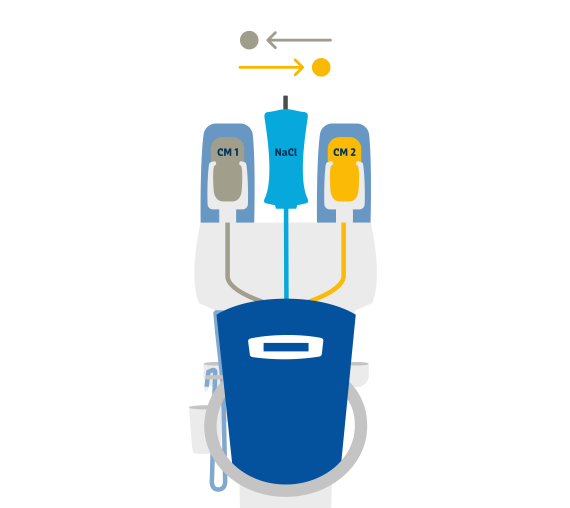
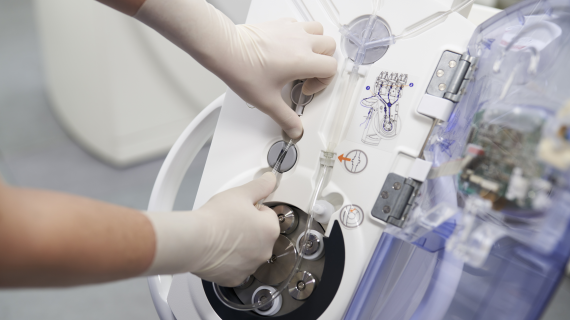
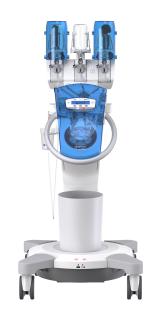
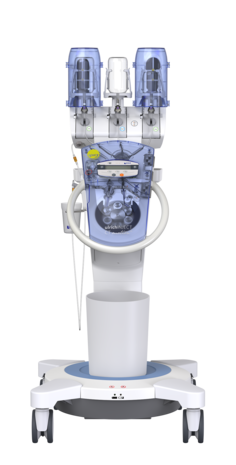
Efficient injection of contrast media for CT with CT motionTM
How can I ensure a reliable, stress-free and cost-effective diagnosis despite time pressure in CT?
Day-to-day work in CT is often defined by time pressure, which makes extensive and personal patient support difficult. Ideally, the right technical equipment can simplify your working processes with as few steps as possible required.